Problem 22.3-2: The Wet and Dry Bulb Psychrometer
The system, shown in Fig. 22.3-2, is a pair of thermometers,
one of which is covered with a cylindrical wick kept saturated with water.
The wick will cool by evaporation into the moving air stream and for
steady operation will approach an asymptotic value know as the wet bulb temperature. The bare
thermometer, on the other hand, will tend to approach the actual temperature of
the approaching air, and this value is called the dry bulb temperature.
Develop an expression for determining the humidity of the air from the
wet and dry bulb temperature readings neglecting radiation and assuming that
the replacement of the evaporating water has no significant effect on the wet
bulb temperature measurement.

Assumptions:
1) Fluid velocity is high enough that reading is unaffected
by radiation and heat conduction along the thermomenter stems but not too fast
where viscous dissipation heat effects are insignificant.
2) Heat of vaporization can be considered the difference
between the heat at the surface of the wick and the heat at the surface of the
thermometer.
Definition of Symbols:
c = concentration
Cphat = heat capacity per unit mass
Cptil = heat capacity per mole
DAB = diffusion coefficient
dHvap = heat of vaporization
Dia = diameter of thermometer with wick
hm =heat transfer coefficient
kxm = mass transfer coefficient
L = length of the wick
NA0 = molar flux for species A
Num = Nusselt number
Pr = Prandtl number
Shm = Sherwood number
Tinf = dry bulb temperature
T0 = wet bulb temperature
WA0 = molar flow rate across the surface
xAinf = mole fraction of water in the approaching air
xA0 = initial film water concentration
|
> |
restart; |
Starting with the energy balance
|
> |
eqn22_3_32:=Q=WA0*(HbarA1-HbarA0); |
![]()
The area corresponds to a cylinder (the shape of
the thermometer)
|
> |
A:=(Dia,L)->Pi*Dia*L; |
![]()
|
> |
Q:=hm*A(Dia,L)*(Tinf-T0); |
![]()
Replacing the change in enthalpy with dHvapA
|
> |
HbarA1:=HbarA0+dHvapA; |
![]()
|
> |
eqn22_3_32; |
![]()
To determine the desired quantity, we must solve the
equation of continuity for this system.
Simplification of:
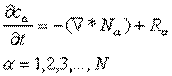
for steady, one-dimensional transport, in the absence of
chemical reactions and external forces, gives:
|
> |
Fluxeqn:=y->D(Nay)(y)=0; |
![]()
Therefore NAy is constant throughout the
wick. To determine the mole
fraction profile, we need the molar flux for diffusion of A through stagnant B:
|
> |
NAy:=y->-c*DAB*D(xA)(y)/(1-xA(y)); |
![]()
Insertion of the above equation into the equation of
continuity and integration gives the mole fraction profile:
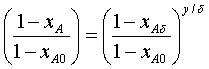
Here we have taken cDAB to be constant, at the
value for the mean wick temperature.
We can then evaluate the constant flux NAy from the preceding
two equations:
|
> |
NA0:=-c*DAB*(xAinf-xA0)/(1-xA0); |
![]()
From the definition of the mass transfer coefficient:
|
> |
WA:=A(Dia,L)*NA0; |
![]()
Rearranging the equation gives
|
> |
eqn22_3_33:=WA0*(1-xA0)=kxm*A(Dia,L)*(xA0-xAinf); |
![]()
|
> |
s:=solve({eqn22_3_32,eqn22_3_33},{WA0,hm}); |
![]()
|
> |
assign(s); |
|
> |
simplify(hm/kxm/dHvapA); |
![]()
Thus we have verified eq. 22.3-34
|
> |
simplify(%-(xA0-xAinf)/(Tinf-T0)/(1-xA0)); |
![]()
Combining equations 22.3-32 and 22.3-33 gives
the following equation
|
> |
hm:='hm':
eqn22_3_34:=hm/kxm/dHvapA=(xA0-xAinf)/(Tinf-T0)/(1-xA0); |
![]()
We then transform the left hand side into
dimensionless form. In the conversion between the heat capacity per mass
(Cphat) to the heat capacity per mol (Cptil), we assume the concentration c =
1.
|
> |
hm:=Num*k/Dia;kxm:=Shm*DAB/Dia;k:=Cphat*mu/Pr;DAB:=mu/rho/Sc;Cphat:=c*Cptil/rho;
c:=1: eqn22_3_34; |
![]()
![]()
![]()
![]()
![]()
![]()
|
> |
Num:=F(Reynolds)*Pr^n; |
![]()
|
> |
Shm:=F(Reynolds)*Sc^n; |
![]()
This simplifies to the following equation
(22.3-38)
|
> |
eqn22_3_38:=simplify(eqn22_3_34); |
![]()
Now evaluating this equation with numbers:
If an air-water mixture at 1 atm pressure gives a wet bulb
temperature of 70 F and a dry bulb temperature of 140 F then
From Dr. Davis' start301
Matlab program vappr with
Tdeg=F and H2O as the only compound:
>>vappr(70)
ans =
2.5172 kPa
|
> |
pAvap:=0.0247*atm;
p:=1*atm; xA0:=pAvap/p; T0:=70*F; Tinf:=140*F; |
![]()
![]()
![]()
![]()
![]()
From Dr. Davis' FORTRAN program steam
Steam; given p,t,x
p = 14.700
psia
s = 0.07456 Btu/lbm-R
t =
70.000 F
h
= 38.09 Btu/lbm
v =
0.16049E-01 ft3/lbm u = 38.05
Btu/lbm
Compressed liquid
Steam; given p,t,x
p = 0.363
psia s
= 2.06449 Btu/lbm-R
t =
70.000 F
h
= 1092.09 Btu/lbm
v =
0.86817E+03 ft3/lbm u = 1033.78 Btu/lbm
Saturated vapor
dH=1092.09-38.09 Btu/lbm and the MW of water is
18.015*lbm/lbmol
|
> |
MW:=18.015*lbm/lbmol:
dHvapA:=(1092.09-38.09)*MW*Btu/lbm; |
![]()
|
> |
Sc:=0.58;
Pr:=0.74;Cptil:=6.98*Btu/lbmol/F; n:=(1/3); |
![]()
![]()
![]()
![]()
|
> |
simplify(eqn22_3_38); |
![]()
The answer we get is slightly higher than that
listed in the example in BSL because the heat of vaporization we used
determined from Dr. Davis' steam
program is 88 Btu/lbm higher than that used in the book.
|
> |
solve(eqn22_3_38,xAinf); |
![]()