Example 17.5-1: Gas Absorption from Rising Bubbles
Bird, Stewart, and Lightfoot. Transport
Phenomena. John Wiley and Sons. 1960.
A Project by Amanda Watford and Judy Hsii
________________________________________________________________________
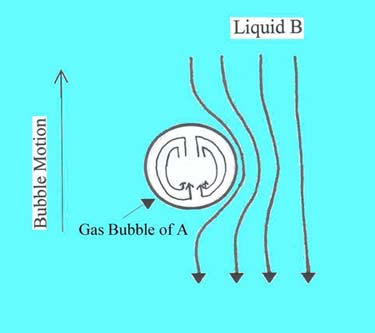
Problem: Gas bubbles of
component A are rising in liquid B at their terminal velocity, Vt,
through a "clean" quiescent liquid.
In this problem, we wish to estimate the rate at which gas A bubbles are
absorbed by liquid B.
________________________________________________________________________
For moderate-sized bubbles in a liquid without
surface-active agent, the bubbles experience a toroidal circulation. Fresh liquid is encountered at the top of
the bubble and the liquid moves downward as the bubble rises. The liquid leaves
as it reaches the bottom of the bubble. Near the interface, the liquid is most
likely laminar flow and keeps its identity. (The liquid behaves similar to
liquid at the surface of a falling film.)
Contact time is deemed to be short as the bubble is traveling at
terminal velocity, so
penetration of dissolved gas A is small.
Therefore, the assumptions utilized in the previous section, Chapter
17.5 Notes, are valid.
Once again, we shall use Maple V to help derive the
solution to the problem. First, all assumptions
will be cleared by using restart.
> restart;
We start with the result obtained in the previous section, Equation
17.5-17 of the BSL text, to estimate the rate of gas absorption and the change
in the bubble size.
> WA := 2*W*cA0*(vmax*L*DAB/Pi)^(1/2);
![]()
WA is the total moles of A transferred per unit time from
the gas to the liquid film. W is the width of the film, cA0 is the solubility
of gas A in liquid B, vmax is the terminal velocity, and L is the length of the
film.
Mass transfer is directly proportional to the square root
of the diffusivity and is inversely proportional to the square root of the
exposure time. t = L/vmax. We use this
relation to define vmax.
> vmax:=L/t;
![]()
After defining vmax, WA is now changed to include this
definition.
> WA;
![[Maple Math]](images/1751example3.gif)
We now designate WA as a positive value and ask Maple to
simplify the expression.
> WA:=simplify(WA,assume=positive);
![[Maple Math]](images/1751example4.gif)
We are interested in the average rate of mass transfer of
material A. Thus, dividing WA, the
total moles of A transferred per unit time from gas A to the liquid, by the
width and length, W and L, respectively, we are left with NAavg, the
average rate of mass transfer of A. The equation shown below is comparable to
the equation shown as Equation 17.5-20 of the BSL text.
> NA[avg]:=(WA/W/L);
![[Maple Math]](images/1751example5.gif)
To verify that the equations are the same, we will
subtract the equation the text gives with the one derived. By inspection, the expression is equal to
zero.
> difference:=simplify(NA[avg]-(4*DAB/Pi/t)^(1/2)*cA0,assume=positive);
![]()
Maple sometimes has a hard time simplify things that
inherently can be simplified. Thus, we will expand the expression above, and
then simplify it to show that the difference is in fact zero.
> expand(%);
![[Maple Math]](images/1751example7.gif)
> simplify(%);
![]()
The time required for liquid B to slide from the top of
the bubble to the bottom of the bubble is the exposure time. For the gas bubble, D is the diameter. Thus, for moderate bubbles of 0.3-0.5 cm in
diameter, the exposure time can be approximated as t‚D/vt, where vt is the
terminal velocity.
> t:=D/vt;
![]()
After defining the exposure time, NAavg
simplifies to the expression given below.
> NA[avg]:=eval(simplify(NA[avg]));
![[Maple Math]](images/1751example10.gif)
We now desire to compare it to the equation derived in
the text for the absorption rate through the bubble-liquid interface, Equation
17.5-21. Once again, we will subtract
the equation given in the text from the one derived above. If the equations are the same, then the
difference should be zero.
> differ:=simplify(NA[avg]-(4*DAB*vt/Pi/D)^(1/2)*cA0,assume=positive);
![[Maple Math]](images/1751example11.gif)
Again, Maple has a difficult time simplifying an
expression that is inherently simplifiable. We will attempt to expand the
expression then simplifying it see if the difference is zero.
> expand(%);
![[Maple Math]](images/1751example12.gif)
> simplify(%);
![]()
The difference between the equations derived using Maple
and the one the book gives as the solution, Equation 17.5-21, is zero. Thus,
the two equations are the same. The
rate through the bubble liquid interface is verified.